
Comparative trial of lung lesions associated with Mycoplasma hyopneumoniae after vaccinating with MHYOSPHERE® PCV ID
Mycoplasma hyopneumoniae (Mhyo) is the primary agent involved in enzootic pneumonia (EP) and one of the leading agents involved in porcine respiratory disease complex (PRDC). EP carries a highly significant economic burden as it reduces average daily weight gain, raises the conversion rate and increases the number of days in the fattening phase. Hence, its management is essential for reducing costs caused by the disease on pig farms.1
Introduction
The objective of the study was to compare lung lesions consistent with Mhyo at the slaughterhouse on several fattening farms in pigs with the same farrowing origin before and after using MHYOSPHERE® PCV ID.
Materials and methods
Between March and July 2021, the lungs of 10 batches of pigs from different fattening farms with the same farrowing origin were evaluated in the slaughterhouse.
The first two batches (387 pigs) were given Vaccine A (an intramuscular vaccine against Mhyo), and the next eight batches (1,759 pigs) were vaccinated with MHYOSPHERE® PCV ID (an intradermal vaccine against Mhyo and PCV2, all in one) which was administered using the HIPRADERMIC® intradermal device.
Apart from the change in vaccine, there were no other notable changes in management or farm fattening facilities.
The following parameters for the lung lesions in the slaughterhouse were evaluated: lesion incidence (lungs [%] with lung lesion), average lesion (average lesion grade among all lungs) and lesion rate (average lesion grade among all affected lungs).
The modified Madec system was used to evaluate the lung.2,3. Statistical analysis was performed using the R software program.
Results
The incidence of Mhyo-compatible lesions were reduced from 51.68% with vaccine A to 32.92% with MHYOSPHERE® PCV ID (p=0.008) (Figure 1), a reduction of 36%. The mean lesion went from 1.01 to 0.45 (p=0.009), a reduction of 55%, and the lesion rate went from 1.96 to 1.38 (p=0.002), a reduction of 29%.
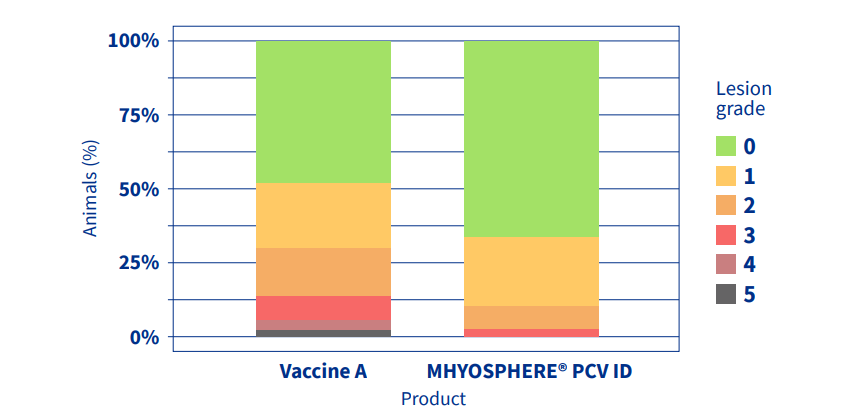
Figure 1. Distribution of animals by lesion grade and product used, and lesion
incidence (animals [%] with lesion [grades 0-5]).
After starting to use MHYOSPHERE® PCV ID, it can be seen chronologically (Figure 2) how the incidence of Mhyo-associated lung lesions is significantly reduced and maintained over the months.
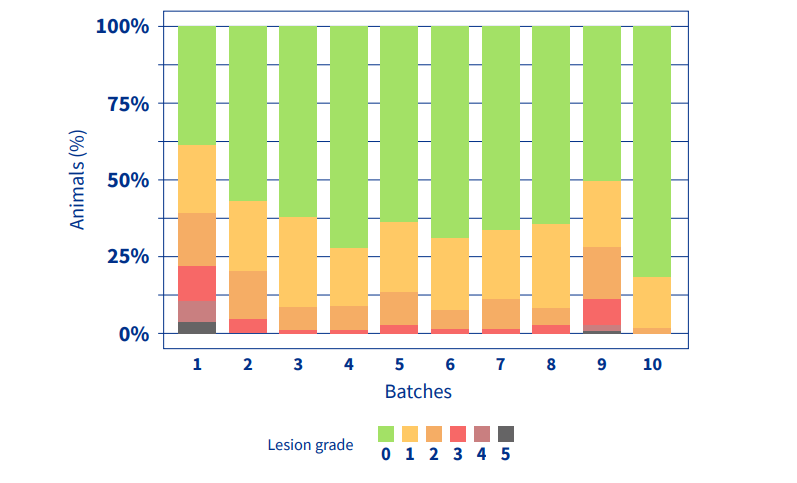
Figure 2. Distribution of animals by lesion grade and batch. Batches 1-2
(Vaccine A), batches 3-10 (MHYOSPHERE® PCV ID).
Conclusions
The new intradermal vaccine against Mhyo and PCV2, MHYOSPHERE® PCV ID, significantly reduced the incidence and mean number of Mhyo-lesions up to the time of slaughter compared to the previous treatment, which could consequently reduce the economic costs associated with this disease.
References
1- Pieters M. and Maes D. Chapter Mycoplasmosis (863-883) Diseases of Swine, Eleventh Edition. Edited by Jeffrey J. Zimmerman et al. (2019) John Wiley & Sons, Inc. Published 2019 by John Wiley & Sons, Inc.
2- Christensen G, Sorensen V, Mousing J (1999) Diseases of the respiratory system. In: Diseases of Swine, 8th Edit., B Straw, SD ‘Allaire, W Mengeling, DJ Taylor, Eds., Iowa State University Press, Ames, pp. 913e940.
3- Madec F, Kobisch M (1982) Bilan lesionnel des poumons de porcs charcutiers à l’abattoir [Assessment of lung lesions in fattening pigs at the slaughterhouse]. Journees de la Recherche Porcine [Swine Research Conference], 14, 405e412.

